Mental Health and Cognitive Sciences
The Mental Health and Cognitive Sciences Unit hosts a team of trained and experienced psychologists and public health researchers. The team conducts community-based randomized controlled trials. The research conducted primarily aims at understanding ways to accelerate development in children as well as addresses mental health issues in mothers, young children, and adolescents. The team is also working on newer tools to measure neurodevelopment in children such as eye tracking assessment and global scale for early development (GSED).
Zinc Supplementation and Mental and Psychomotor Development

In this double-blind, randomized, placebo-controlled trial, children aged 6 to 30 months received daily
elemental zinc (10 mg for infants and 20 mg for others) or placebo for 4 months. Bayley Scales of Infant Development II were used for development assessment in the 12- to 18-month subgroup at enrollment and the end of the study. Zinc supplementation did not affect the mental or psychomotor development index scores in a setting in which zinc deficiency is common (J Pediatr 2005;146(4):506-11).
Cobalamin and Folate Status Predicts Mental Development Scores
The objective of the study was to measure the association of plasma concentrations of folate, cobalamin, total homocysteine, and methylmalonic acid with cognitive performance at 2 occasions, 4 mo apart, in North Indian children aged 12–18 mo. Cobalamin and folate status showed a statistically significant association with cognitive performance. Given the high prevalence of deficiencies in these nutrients, folate and cobalamin supplementation trials are required to measure any beneficial effect on cognition (Am J Clin Nutr. 2013 Feb;97(2):310-7).

Assessment of Developmental Status using the Ages and Stages Questionnaire-3 in Nutritional Research
For large epidemiological studies in low and middle-income countries, inexpensive and easily administered developmental assessment tools are called for. We evaluated the feasibility of the assessment tool Ages and Stages Questionnaire 3.edition (ASQ-3) “home procedure” in a field trial in 422 North Indian young children. We found that the translated and adjusted ASQ-3 “home procedure” was a feasible procedure for the collection of reliable data on the developmental status in infants and young children (Nutr J. 2013 Apr 23;12:50. doi: 10.1186/1475-2891-12-50).
Diarrhea, Stimulation and Growth Predict Neurodevelopment
Infants and young children in low to middle-income countries are at risk for adverse neurodevelopment due to multiple risk factors. In this study, we sought to identify stimulation and learning opportunities, growth, and burden of respiratory infections and diarrhea as predictors for neurodevelopment. Our results support the importance of early child stimulation and general nutrition for child development. Our study also suggests that diarrhea is an additional risk factor for adverse neurodevelopment in vulnerable children (PLoS One. 2015;10:e0121743).

Cognitive and Motor Outcomes in Children Born Low Birth Weight: A Meta-analysis

This meta-analysis was conducted on the premise that synthesized evidence is lacking on magnitude of cognitive and motor deficits in low birth weight (LBW i.e. birth weight< 2500 g) children compared to those with normal birth weight (NBW) in South Asian context. A total of 19 articles (n = 5999) were included in the analysis. Children < 10 years of age born LBW had lower cognitive (WMD -4.56; 95% CI: -6.38, – 2.74) and motor scores (WMD -4.16; 95% CI: -5.42, – 2.89) compared to children with NBW. Within LBW children, those with birth weight < 2000 g had much lower cognitive (WMD -7.23, 95% CI; – 9.20, – 5.26) and motor scores (WMD -6.45, 95% CI; – 9.64, – 3.27). The review concluded that in south Asia, children born LBW, especially with < 2000 g birth weight, have substantial cognitive and motor impairment compared to children with NBW (BMC Pediatr 2019 Jan 29;19(1):35)
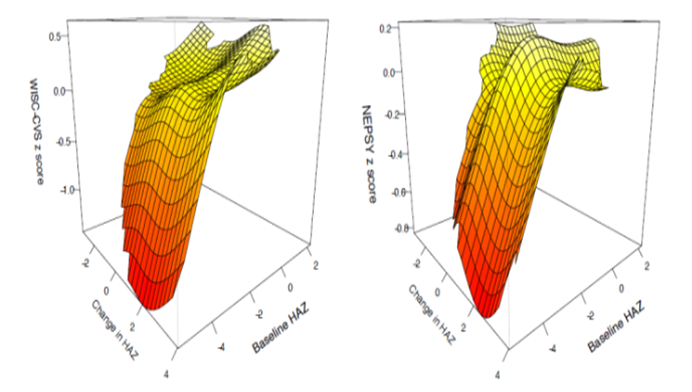
Linear Growth between Early and Late Childhood and Cognitive Outcomes at 6-9 Years of Age
In a follow-up study of a placebo-controlled trial among children aged 6-30 months, we measured growth and neurodevelopment when the children were 6–9 years old. The objective was to assess the extent to which improvements in linear growth between early (12-36 months) and late (6-9 years) childhood impact cognitive outcomes at 6-9 years of age. Neurodevelopment was assessed using standardized and validated psychometric tools (Wechsler Intelligence Scale for Children, 4th edition; Crichton Vocabulary Scales; and Neuropsychological test battery). Height for age z scores (HAZ) in the first 2 years of life were significantly associated but there were no significant associations between change in HAZ between early and later childhood and neurodevelopmental outcomes. The findings support the current practice of investing efforts to accelerate linear growth in the first 2-3 years of life (J Pediatr. 2020 ;225:214-221.e3).
Vitamin B-12, Folate, and Cognition

Vitamin B12 and folate are important for normal brain development. The objective of this study was to measure the effects of 6-month supplementation of vitamin B12 and/or folic acid in early childhood on cognition when the children were 6 to 9 years old. The findings, from both an observational and a randomized design, suggested that vitamin B12 and folate in children 6 to 36 months have limited public health relevance for long-term cognition (Pediatrics 2020;145(3):e20192316).
Association between Vitamin D Status and Cognitive Development and Linear Growth
In a follow-up study of a placebo-controlled trial among children aged 6-30 months, we measured growth and neurodevelopment when the children were 6–9 years old. Neurodevelopment was measured using the Wechsler Intelligence Scale for Children, 4th edition INDIA, the Crichton Verbal Scale, NEPSY-II subtests, and the BRIEF 2. Vitamin D status was not associated with any of the cognitive outcomes or linear growth. Our findings did not support the notion that poor vitamin D status in early childhood is an important limitation for cognitive development and linear growth (Nutr J 2020;19(1):14).

Community-initiated Kangaroo Mother Care and Early Child Development

We conducted a randomized controlled trial among LBW infants, majorly late preterm or term small for gestational age infants without any problems at birth and weighing 1500–2250 g at birth. The intervention comprised of promotion of skin-to-skin contact and exclusive breastfeeding by trained intervention delivery team through home visits. The intervention group mother-infant-dyads were supported to practice ciKMC till day 28 after birth or until the baby wriggled-out. All infants in the intervention and control groups received Home Based Post Natal Care visits by government health workers. Cognitive, language, motor and socio-emotional outcomes were assessed at infant-ages 6- and 12-months using Bayley Scale of Infant Development (BSID-III). Other outcomes measured were infant temperament, maternal depression, maternal sense of competence, mother-infant bonding and home-environment. Our study was unable to capture any effect of ciKMC on neurodevelopment during infancy in this sample of stable late preterm or term small for gestational age infants. Long term follow-up may provide meaningful insights (BMC Pediatr. 2020;20(1):150).
Prebiotic and Probiotic Supplementation on Neurodevelopment in Preterm Very Low Birth Weight
Preterm very low birth weight (VLBW) infants are at risk of gut dysbiosis and neurodevelopmental deficits. Prebiotics and probiotics may modulate gut microbiota and influence brain functions. This review synthesized literature on effect of prebiotic and/or probiotic supplementation in preterm VLBW on their neurodevelopmental outcomes. Out of 275 articles identified, seven randomized controlled trials (RCTs) were included for review. Age of assessment of outcomes was ≥18-22 months of corrected age in included studies. The meta-analysis found that supplementation with prebiotic and probiotics did not decrease or increase the risk of cognitive and motor impairment, cerebral palsy, visual, and hearing impairment. Quality of evidence was “low” to “very low.” The meta-analysis concluded that limited evidence from RCTs does not demonstrate a difference in neurodevelopmental outcomes between prebiotic/probiotic treated and untreated control groups (Pediatr Res. 2020;87(5):811-822).

Use of the Global Scale for Early Development (GSED) within the Women and Infants Integrated Growth Study (WINGS) for Impact Evaluation on Neurodevelopment

The GSED tool has been developed by WHO with the intent of creating a harmonized scale to measure child development in children under 3 years. The GSED tools are currently being validated in a cohort of children in several countries. However, it will still be unknown how the GSED tools perform in an intervention trial setting, specifically, in terms of their ability to detect intervention effects on developmental outcomes.
The proposed study in 1500 infants aims to use the ongoing WINGS as a platform to test the GSED tools. It will also provide an opportunity to understand how the responsiveness of GSED tools compare to standardized psychometric tools (Ages and Stages Questionnaire and Bayley Scales of Infant and Toddler Development) in detecting intervention effects on neurodevelopmental outcomes. The specific objectives are:
– To evaluate the ability of the GSED instruments in detecting effects of the integrated interventions on child neurodevelopmental outcomes at 6, 12 and 24 months of age in a setting where the intervention is likely to improve linear growth
– To determine predictive validity of GSED (measured at 12 months of age) for neurodevelopmental outcomes measured at 24 months of age
Status: Study ongoing (Trial Registration CTRI/2020/08/027228)
Nutritional Interventions During Infancy to Improve Neurodevelopmental Outcomes
Paucity of data from randomized controlled trials in resource-constrained settings limits the scientific understanding on the effect of supplementing balanced protein-energy complementary feeds enriched with micronutrients during infancy on neurodevelopment.
We are conducting a study to examine the effect of supplementing infants from resource-limited settings in India with improved quality of complementary feeds on their neurodevelopment with the following objectives:
To test in 1290 infants, the effect of two food supplements given daily for 6 months, starting from 6 months of infant age, that vary in total protein amount (2.5 g protein; protein energy ratio of 8% vs 5.6 g protein; protein energy ratio of 18%) and amount of protein from dairy source (0.75 g vs 1.68 g) but similar in energy (125 kcal), fat (30-45% of total energy) and micronutrient content (1 RDA) on the Bayley Scales of Infant and Toddler Development, 3rd Edition (BSID-III) cognitive, language and motor scores at 12 and 24 months of age in comparison to standard care (counselling with no food supplementation).
To compare the effects of the two food supplements given daily for six months, starting from 6 months of infant age, against standard of care on socio-emotional functioning, infant temperament and behavioural problems at 12 and 24 months of age. Status: Study Ongoing (Trial Registration CTRI/2020/08/027228)

Assessment of Eye Tracking During Infancy as a Tool to Detect Developmental Deficits in Young children at 24 Months of Age
Existing techniques for assessing early cognitive development in children are limited by the requirement for manual test administration, subjective judgements of infant behaviour and lack of sensitivity to specific neurocognitive processes. It is important to develop objective assessment techniques for early identification of children at risk for long-term cognitive deficits.
Use automated tracking of eye movements in infants and children aged under 24 months of age to obtain eye tracking parameters at specific time points and attempt to understand the strength of association of the parameters to neurodevelopmental outcomes at 24 months of age, assessed by Bayley Scales of Infant and Toddler Development (BSID).
Findings will be helpful to understand the feasibility of conducting automated visual tracking of infants and children in community-based settings. Status: Approved for funding.
Long Term Neurodevelopment Outcomes of Low Birth Weight Infants Receiving Kangaroo Mother Care

Low birth weight infants are at a high risk of impaired cognitive performance
KMC carries a strong biological plausibility of improving neurodevelopment through ensuring optimal nutrition and stimulation, improving maternal responsiveness and mother-infant interaction during the critical window of rapid brain growth
The current evidence on the impact of KMC on long term development is limited
The proposed study will be an extended follow up of LBW infants enrolled in a community based randomized controlled trial with the intent to assess the effect of KMC during neonatal period on cognitive, language, socio-emotional, higher executive and early academic skills scores at ages 6-7 years. Status: To be initiated soon (Wellcome-DBT India Alliance Early Career Fellowship, Ravi Upadhyay, Young Scientist)
